Real-World Evidence in the Drug Approval Process
This article explores the growing role of RWE in drug approvals, its benefits, challenges, and future implications for the pharmaceutical industry.

Introduction
In the evolving landscape of drug development, Real-World Evidence (RWE) has emerged as a critical component in regulatory decision-making. Traditionally, randomized controlled trials (RCTs) are the gold standard for evaluating drug efficacy and safety. However, RCTs can be expensive, time-consuming, and may lack representation of real-world patient populations.
With advancements in big data analytics, artificial intelligence (AI), and regulatory policy changes, pharmaceutical companies and regulatory agencies are increasingly incorporating RWE to supplement clinical trial data. This shift is accelerating drug approvals, expanding label indications, and ensuring post-market safety monitoring.
This article explores the growing role of RWE in drug approvals, its benefits, challenges, and future implications for the pharmaceutical industry.

What is Real-World Evidence (RWE)?
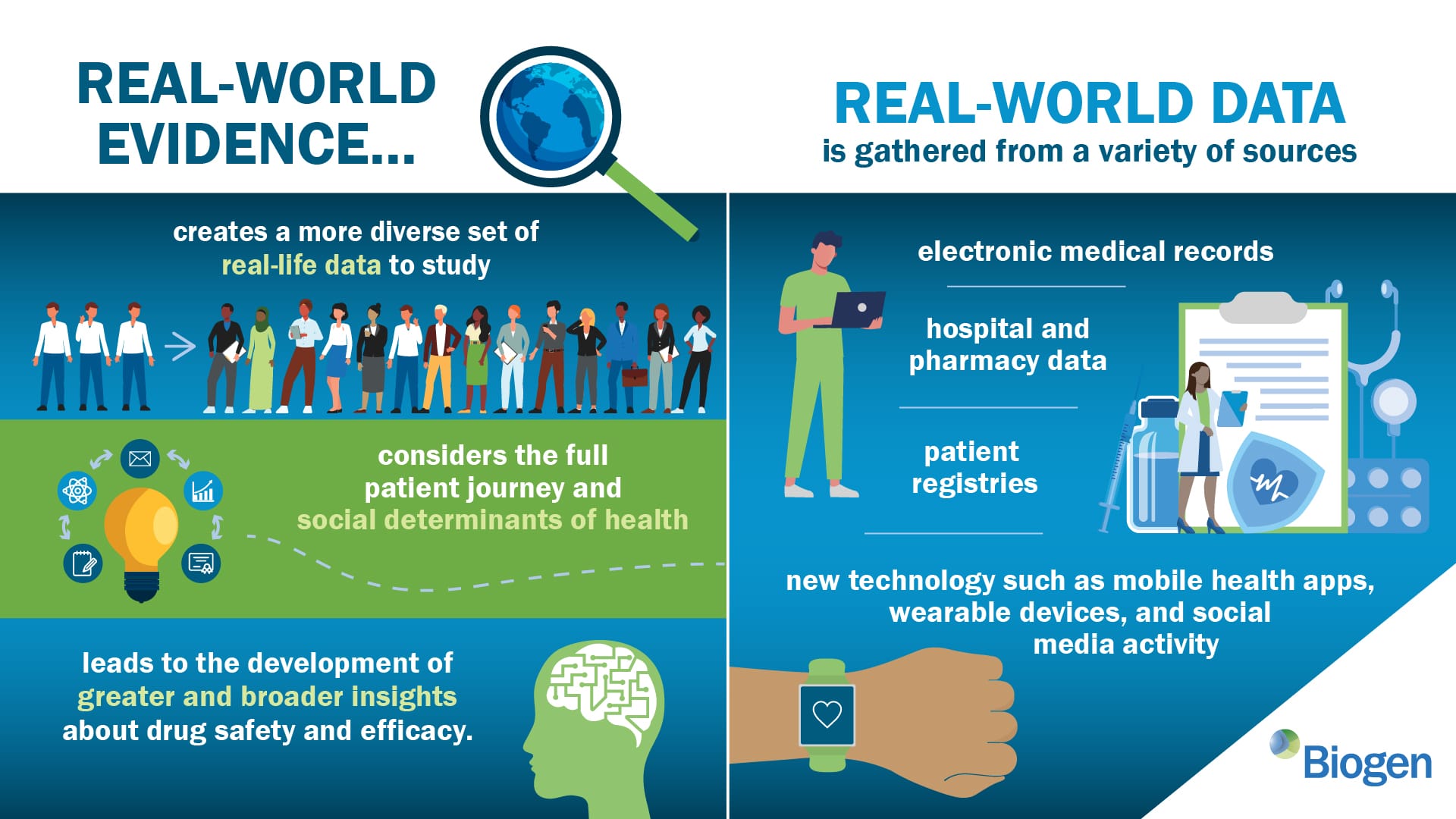
Real-World Evidence (RWE) refers to clinical evidence derived from real-world data (RWD) that reflects patient experiences outside of controlled clinical trials. It provides insights into how a drug performs in broader, everyday settings.
Sources of RWD include:
- Electronic Health Records (EHRs): Patient medical histories, laboratory results, and physician notes.
- Insurance Claims & Billing Data: Patterns of drug use and healthcare service utilization.
- Patient Registries & Disease Databases: Information from patients with specific diseases.
- Wearable Devices & Mobile Health Apps: Continuous monitoring of patient health metrics.
- Social Media & Patient-Reported Outcomes: Unfiltered real-time feedback from patient communities.
By leveraging these sources, RWE enhances traditional clinical trial data, providing a more comprehensive picture of a drug's long-term safety and effectiveness.
Why is Real-World Evidence Gaining Importance?
1. Limitations of Traditional Clinical Trials
RCTs, while essential, have several drawbacks:
- Expensive & Time-Consuming: The average drug trial takes 7-10 years and costs billions of dollars.
- Controlled Populations: Excludes many real-world patients (e.g., elderly, pregnant women, those with comorbidities).
- Small Sample Sizes: May not capture rare adverse events that emerge in broader populations.
2. Regulatory Evolution
Regulatory agencies are adapting to the need for RWE:
- FDA’s 21st Century Cures Act (2016) encouraged the use of RWE in drug approvals.
- The European Medicines Agency (EMA) and other global regulators are increasingly integrating RWE into decision-making.
3. Faster Drug Approvals for Unmet Medical Needs
For diseases like cancer, rare genetic disorders, and COVID-19, waiting for RCTs can delay access to life-saving treatments. RWE enables faster regulatory pathways, particularly for orphan drugs and breakthrough therapies.
4. Advancements in AI & Big Data Analytics
AI-driven tools can extract meaningful patterns from vast amounts of RWD, improving data quality, speed, and reliability in evidence generation.

How Is RWE Being Used in Drug Approvals?
RWE is transforming pharmaceutical regulatory processes in several ways:
✅ Post-Market Surveillance: Identifies rare side effects and long-term safety concerns after a drug is approved.
✅ Label Expansion: Used to approve new indications for existing drugs (e.g., expanding cancer therapies to additional tumor types).
✅ Accelerated Approval Pathways: Supports conditional approvals for drugs treating high unmet needs.
✅ Comparative Effectiveness Research: Evaluates how a drug performs against existing treatments in real-world settings.

Case Studies:
- Pfizer & Moderna’s COVID-19 Vaccines: RWE confirmed use in real-world populations, leading to expanded emergency use approvals.
- Ibrance (palbociclib) for Breast Cancer: FDA accepted RWE to expand its indications, validating its benefits beyond clinical trial participants.
Benefits of Incorporating RWE in Drug Approvals
✅ Faster Access to Life-Saving Medications – Reduces the time required for regulatory approvals.
✅ More Inclusive Patient Data – Captures real-world patient diversity, including populations often excluded from RCTs.
✅ Cost Savings for Pharma & Patients – Streamlines drug development, lowering overall healthcare costs.
✅ Improved Post-Market Safety Monitoring – Allows for ongoing assessment of adverse effects.
✅ Enhancing Personalized Medicine – Enables tailored treatment strategies based on real-world genetic and behavioral data.
Challenges and Limitations of RWE
Despite its benefits, RWE presents several challenges:
❌ Data Quality & Reliability Issues:
- RWD is often incomplete, inconsistent, or biased due to different data collection methods.
- Lack of standardization across health records can make comparisons difficult.
❌ Regulatory & Ethical Concerns:
- Privacy risks associated with collecting and analyzing large-scale patient data.
- Unclear regulatory frameworks for determining when RWE is sufficient for approval.
❌ Potential Bias in Data Collection:
- RWE studies may be influenced by confounding factors, leading to incorrect conclusions.
- Patients using wearables or digital health tools may not represent the general population.
❌ Pharmaceutical Industry Influence:
- Risk of misuse if companies selectively use RWE to support favorable conclusions while ignoring negative findings.

The Future of RWE in Drug Development
🔹 Regulatory Frameworks Will Continue to Evolve – Expect stronger guidelines for RWE collection and use in approvals.
🔹 AI & Machine Learning Will Enhance Data Quality – Advanced analytics will improve RWE accuracy and predictive capabilities.
🔹 Greater Use in Personalized Medicine – RWE will play a crucial role in precision medicine, tailoring treatments to individual patient needs.
🔹 More Pharma-Led RWE Studies – Companies will proactively generate RWE to support both regulatory submissions and market adoption.
Conclusion
The integration of Real-World Evidence into drug approval processes marks a significant shift in the pharmaceutical industry. By complementing traditional clinical trials with real-world data, regulators and drug developers can make faster, more informed decisions that improve patient outcomes and access to innovative therapies.
However, ensuring data reliability, regulatory alignment, and ethical considerations will be critical to unlocking RWE’s full potential. As technology and data analytics continue to advance, RWE is set to become an indispensable tool in the future of drug development, regulatory science, and precision medicine.
References:
1) Using real-world evidence to supplement randomized controlled trials and reduce health inequity. STAT. https://www.statnews.com/sponsor/2022/03/18/using-real-world-evidence-to-supplement-randomized-controlled-trials-and-reduce-health-inequity/
2) ArborAdmin. 9 Ways Real-World Evidence is Changing Healthcare. ArborMetrix. Published August 9, 2021. https://www.arbormetrix.com/blog/9-ways-real-world-evidence-is-changing-healthcare/
3) Real-World Evidence | Prospective Studies | Retrospective Studies | AmerisourceBergen. Amerisourcebergen.com. Published 2025. Accessed April 2, 2025. https://www.amerisourcebergen.com/manufacturer-solutions/health-economics-market-access-regulatory-consulting/real-world-evidence
4) Real-World Evidence And The Quest For European Market Access. Insights. Published November 22, 2016. Accessed April 2, 2025. https://insights.citeline.com/IV004954/Real-World-Evidence-And-The-Quest-For-European-Market-Access/
*Information presented on RxTeach does not represent the opinion of any specific company, organization, or team other than the authors themselves. No patient-provider relationship is created.

